These 27 eye products could lead to infections and vision loss, FDA warns
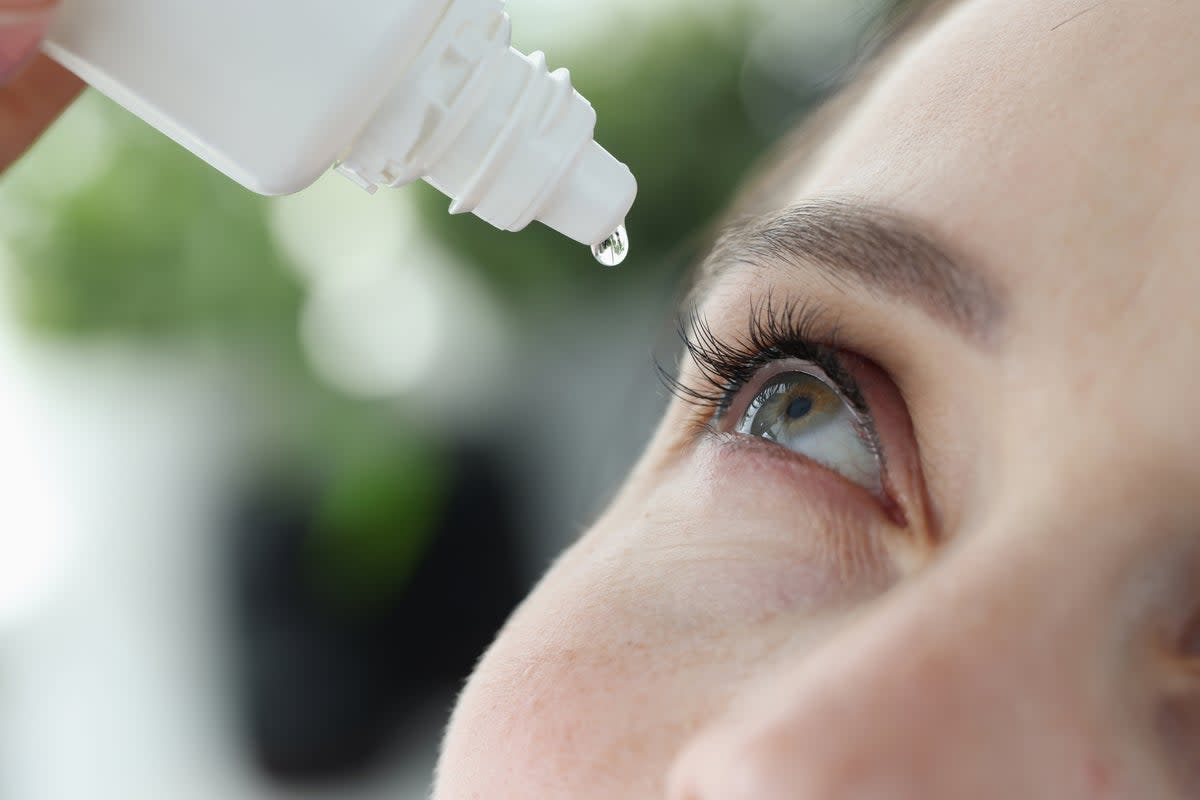
The US Food and Drug Administration (FDA) is warning consumers not to use several over-the-counter eye drops that may be dangerous.
The agency issued a statement Friday listing 26 products that should not be used, and added a 27th to the list on Monday.
Many of the products are available at popular chain pharmacies such as CVS and Rite Aid, and others have been sold at Target and Walmart.
“FDA is warning consumers not to purchase and to immediately stop using [these products] due to the potential risk of eye infections that could result in partial vision loss or blindness,” the agency’s statement said.
If you have over-the-conter eye drops in your home, you should check the list provided by the FDA to make sure they aren’t among those that have been affected. It includes common brands such as Equate Hydration PF Lubricant Eye Drops and many different Lubricant products.
As of 30 October, there hadn’t been any reports of eye infections associated with the products; however, the FDA said the conditions in which they were produced made them unsafe. “Agency investigators found insanitary conditions in the manufacturing facility and positive bacterial test results from environmental sampling of critical drug production areas in the facility,” the statement said.
It went on to explain why compromised eye products can be so dangerous to consumers: “These products are intended to be sterile. Ophthalmic drug products pose a potential heightened risk of harm to users because drugs applied to the eyes bypass some of the body’s natural defenses,” the statement said.
In addition to warning the public, the agency “recommended” that the manufacturers of the compromised products issue formal recalls.
The affected products sold at Target, Walmart, Rite Aid, and CVS have been removed from store shelves and websites.
Anyone who develops symptoms of an eye infection and has recently used one of the affected products should “seek medical care immediately”, per the FDA.
The following are all possible signs of an eye infection, per the Cleveland Clinic: irritation, itching, eye pain, red eyes, watery eyes, swelling, discharge (that may be yellow or sticky) coming from the eyes, sensitivity to light (also known as photophobia), fever, blurred vision, and noticing that one’s eyelashes are sticking together because of discharge.
An eye infection can be diagnosed after an eye exam, during which a provider may take a swab of discharge coming from the eyes if it is present.
There are multiple treatments for eye infections. Sometimes, antibacterial medications are used, and it can take one to two days for these to kick in. Your eye doctor may also recommend supportive measures such as using a warm damp compress to alleviate discomfort, taking over-the-counter pain relievers, or using lubricating eye drops.
Certain behaviors can lessen your chances of developing an eye infection, including frequently washing your hands with warm water and soap, avoiding people you know are sick, following directions for proper contact lens use, and discarding any cosmetic products that have caused eye irritation in the past.
The new statement from the FDA said that if you find one of the compromised products in your home, you should dispose of it.
If you develop worrisome symptoms after using the product, you can report your injury to the FDA here.


